 |
The Sullivan Lab, MCD Biology, UCSC |

|
|||
Sullivan Lab Research Cytokinesis, Centralspindlin and Vesicle-mediated Membrane Delivery
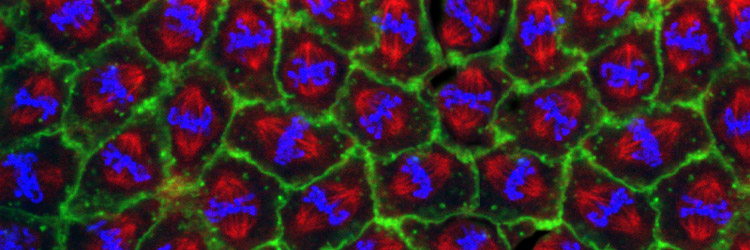
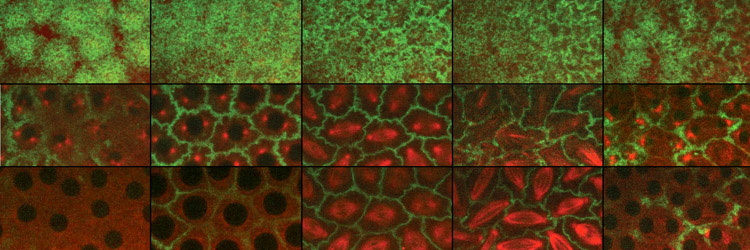
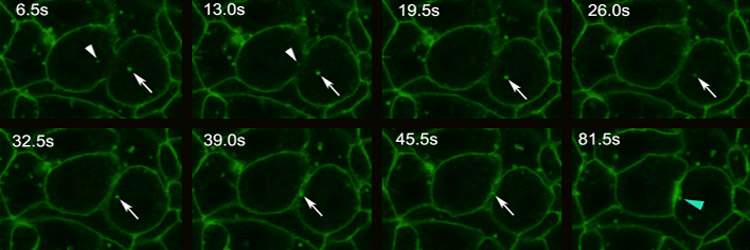
The final event in the mitotic cycle is a dramatic constriction of the plasma membrane, producing two distinct daughter cells. This process, known as cytokinesis, involves formation of an acto-myosin based contractile ring that forms perpendicular and midway to the anaphase spindle. Recent major advances in understanding structural and regulatory mechanisms that guide cytokinesis include the realization that vesicle-mediated membrane addition, as well as acto-myosin based contraction, drives furrow invagination. While it had long been appreciated that Golgi-mediated vesicle fusion drives plant cytokinesis, studies of animal cytokinesis focused on acto-myosin based contraction. However, observational and functional studies from a number of systems revealed that vesicle-based membrane delivery is also an important element of animal cytokinesis. This insight has created new avenues of investigation in the cytokinesis field: what are the sources of membrane, when and where is membrane added during furrow invagination, and how is this process regulated and coordinated with actin-myosin based contraction and the cell cycle. A second insight has been the defining of the role of the central spindle (the overlap microtubules positioned between daughter telophase nuclei) in establishing the site of the cytokinesis furrow. A protein complex consisting of a motor protein MKLP1 and RacGAP known as Centralspindlin, relies on these overlap microtubules to bring a potent actin remodeler RhoGEF to the site of contractile ring formation. Our lab uses a combination of molecular genetic and biochemical approaches to define the source and mechanisms of vesicle-mediated membrane delivery to the contracting furrow. Comprehensive genetic and biochemical screens in our lab have identified the Golgi and Recycling Endosome as important as key sources of membrane for the advancing furrow. We have convicing evidence that Centralspindlin delivers membrane as well as RhoGEF to the site of furrow formation. We are currently testing this by in vivo reconstruction of Centralspindlin movement and on the overlap central spindle microtubules. These experiments enable us to assay known and potential cargoes that associate with Centralsplindlin. Recpresentative Publications: Kotadia S*, Montembault E*, Sullivan W, Royou A. (2012) Cell elongation – an adaptive response clearing long chromatid arms from the cleavage plane. Journal of Cell Biology 199(5):745-53. [PDF] Crest J, Concha-Moore K, and Sullivan W. (2012) RhoGEF and positioning Rappaport-like furrows in the early Drosophila embryo. Curr Biol. 2012 Sep 25. [PDF] Cao J, Crest J, Fasulo B, Sullivan W. (2010) Cortical actin dynamics facilitates early stage centrosome separation. Current Biology 20 (8): 770-776.[PDF] Cao J, Albertson R, Riggs B, Field CM, Sullivan W. (2008) Nuf, a Rab11 effector, maintains cytokinetic furrow integrity by promoting local actin polymerization. J. Cell Biol 182(2): 301-13. [PDF] Albertson R, Cao J, Hsieh TS, Sullivan W. (2008) Vesicles and actin are targeted to the cleavage furrow via furrow microtubules and the central spindle. J Cell Biol. 181(5):777-90. [PDF] For more publications regarding work on cytokinesis, go to our publications page. [Back] [Next] [Sullivan Research Main Page ] |
|||
|
|